The physico-chemical process underlying digestion is called Ostwald ripening. Iron (ll) hydroxide is a green coloured solid. Sodium sulphate Na 2 SO 4 is formed as a co-product. The iron (ll) hydroxide Fe (OH) 2 is an insoluble salt formed when ferrous sulphate FeSO 4 and sodium hydroxide NaOH undergo a reaction. 
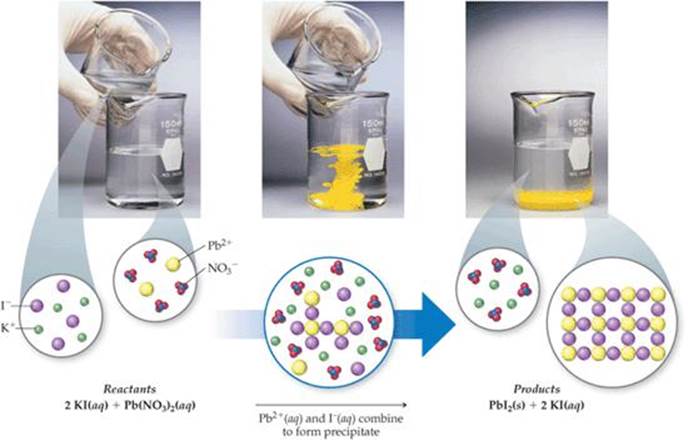
It results in cleaner and bigger particles. Examples of precipitation reactions: Ferrous sulphate and Sodium hydroxide. Similar processes are often used to separate chemically similar elements, such as the rare earth metals.ĭigestion, or precipitate ageing, happens when a freshly-formed precipitate is left, usually at a higher temperature, in the solution from which it is precipitated. Precipitation Reactions: A chemical reaction that involves the formation of an insoluble product (precipitate solid) is called Precipitation reaction. If precipitation is expected, write a balanced net ionic equation for the reaction. To identify the cation, the color of the precipitate and its solubility in excess are noted. Predict the result of mixing reasonably concentrated solutions of the following ionic compounds. To do this, an alkali first reacts with the unknown salt to produce a precipitate which is the hydroxide of the unknown salt. Precipitate formation is useful in the detection of the type of cation in salt. This simplifies the above equations to the following: In this case, any spectator ions (those which do not contribute to the reaction) are left out of the formula completely. This reaction can be written emphasizing the dissociated ions in a combined solutionĪg +(aq) + NO 3 -(aq) + K +(aq) + Cl -(aq) → AgCl(solid) + K +(aq) + NO 3 -(aq)Ī final way to represent a precipitate reaction is known as a net ionic reaction. This can occur when solutions containing ionic compounds. The silver chloride(AgCl) has formed a solid, which is observed as a precipitate. A precipitate is a solid formed in a chemical reaction that is different from either of the reactants. If this energy is not available, and no suitable nucleation surface is available, supersaturation occurs.Īn example of a precipitation reaction: Aqueous silver nitrate (AgNO 3) is added to a solution containing potassium chloride (KCl) and the precipitation of a white solid, silver chloride is observed. The creation of a hypothetical solid particle includes the formation of an interface, which requires some energy based on the relative surface energy of the solid and the solution. Precipitation from a solid solution is also a useful way to strengthen alloys this process is known as solid solution strengthening.Īn important stage of the precipitation process is the onset of nucleation. 
This effect is useful in many industrial and scientific applications whereby a chemical reaction may produce a solid that can be collected from the solution by various methods (e.g. Precipitation reactions can be used for making pigments, removing salts from water in water treatment, and for qualitative chemical analysis. 3 Representation using chemical equations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
